Exam Question for Class 12 Chemistry Chapter 8 The d – and f – Block Elements
Please refer to below Exam Question for Class 12 Chemistry Chapter 8 The d – and f – Block Elements. These questions and answers have been prepared by expert Class 12 Chemistry teachers based on the latest NCERT Book for Class 12 Chemistry and examination guidelines issued by CBSE, NCERT, and KVS. We have provided Class 12 Chemistry exam questions for all chapters in your textbooks. You will be able to easily learn problems and solutions which are expected to come in the upcoming class tests and exams for standard 12th.
Chapter 8 The d – and f – Block Elements Class 12 Chemistry Exam Question
All questions and answers provided below for Exam Question Class 12 Chemistry Chapter 8 The d – and f – Block Elements are very important and should be revised daily.
Very Short Answer Type Questions
Question. Name a member of the lanthanoid series which is well known to exhibit +2 oxidation state.
Answer.Europium (Eu) is well known to exhibit +2 oxidation state due to its half-filled f orbital in +2 oxidation state.
Question. Name a member of the lanthanoid series which is well known to exhibit +4 oxidation state.
Answer.Lanthanoids showing +4 oxidation state are 58Ce, 59Pr, 65Tb.
Question. Zn2+ salts are white while Cu2+ salts are coloured. Why?
Answer.Zn2+ ion has completely filled d-subshell and no d-d transition is possible. So zinc salts are white. Configuration of Cu2+ is [Ar] 3d 9. It has partly filled d-subshell and hence
it is coloured due to d-d transition.
Question. Write the formula of an oxoanion of chromium
(Cr) in which it shows the oxidation state equal to its group number.
Answer.Oxoanion of chromium in which it shows +6 oxidation
state equal to its group number is Cr2O72– (dichromate ion).
Question. Assign reason for the following :
Copper (I) ion is not known in aqueous solution.
Answer.In aqueous solutions, Cu+ undergoes disproportionation to form a more stable Cu2+ ion.
2Cu+(aq) → Cu2+(aq) + Cu(s)
Cu2+ in aqueous solutions is more stable than Cu+ ion because hydration enthalpy of Cu2+ is higher than that of Cu+. It compensates the second ionisation enthalpy of Cu involved in the formation of Cu2+ ions.
Question. Give reasons for the following :
Eu2+ is a strong reducing agent.
Answer.Eu2+ has a strong tendency to lose electrons to attain the more stable +3 oxidation state of lanthanoids hence, it is a strong reducing agent.
Question. Account for the following :
Zn is not considered as a transition element.
Answer.In the electronic configuration of Zn the d-orbitals are completely filled in the ground state as well as in its common oxidation state. So, it is not regarded as transition metal.
Question. Write the formula of an oxoanion of manganese (Mn) in which it shows the oxidation state equal to its group number.
Answer.Formula of oxoanion of manganese is MnO–4.
Oxidation state of Mn in this oxoanion = + 7
Group number of Mn is 7.
Question. Account for the following :
Zr and Hf have almost similar atomic radii.
Answer.Due to lanthanoid contraction the elements of 4d and 5d-series have similar atomic radii e.g., Zr = 145 pm and Hf = 144 pm.
Question. How would you account for the following :
Among lanthanoids, La(III) compounds are predominant. However, occasionally in solutions or in solid compounds, +2 and +4 ions are also obtained.
Answer.Lanthanum and all the lanthanoids predomi-nantly show +3 oxidation state. However, some of the lanthanoids also show +2 and +4 oxidation states in solution or in solid
compounds. This irregularity arises mainly due to attainment of stable empty (4f 0), half-filled (4f 7) and fully filled (4f14) sub shell.
e.g., Ce4+ : 4f 0 , Eu2+ : 4f 7
Tb4+ : 4f 7 , Yb2+ : 4f 14
Short Answer Type Questions (SA-I)
Question. How would you account for the following :
(i) The E°M 2+/M for copper is positive (+0.34 V).
Copper is the only metal in the first series of transition elements showing this behaviour.
(ii) The metallic radii of the third (5d) series of transition metals are nearly the same as those of the corresponding members of the second (4d) series.
Answer.
(i) Electrode potential (E°) value is the sum of three factors :
(a) Enthalpy of atomisation ; ΔaH for Cu(s) → Cu(g)
(b) Ionisation enthalpy ; ΔiH for Cu(g) → Cu2+(g)
(c) Hydration enthalpy ; ΔhydH for Cu2+(g) → Cu2+(aq)
In case of copper the sum of enthalpy of atomisation and ionisation enthalpy is greater than enthalpy of hydration.
This is why E°M2+/M for Cu is positive.
(ii) This is due to lanthanoid contraction.
Question. (i) Write two characteristic of the transition elements.
(ii) Which of the 3d-block elements may not be regarded as the transition elements and why?
Answer.
(i) Elements which have incompletely filled d-orbitals in their ground state or in any one of their oxidation states are called transition elements.
Characteristics of transition elements :
(a) They show variable oxidation states.
(b) They exhibit catalytic properties.
(ii) Zn, Cd, Hg are considered as d-block elements but not as transition elements because they do not have partly filled d-orbitals in their atomic state or their common oxidation states (i.e., Zn2+, Cd2+, Hg2+).
Question. Why is europium (II) more stable than cerium (II)?
Answer.Europium (II) has electronic configuration [Xe]4f 75d 0 while cerium (II) has electronic configuration [Xe] 4f 1 5d1. In Eu2+, 4f subshell is half filled and 5d-subshell is empty. Since,half filled and completely filled electronic configurations are more stable, so Eu2+ ions is more stable than Ce2+ in which neither 4f subshell nor 5d subshell is half filled or completely filled.
Question. Write the electronic configuration of Ce3+ ion, and calculate the magnetic moment on the basis of ‘spin-only’ formula. [Atomic no. of Ce = 58]
Answer.
Ce (Z = 58) = [Xe] 4f1 5d1 6s2
∴ Ce3+ = [Xe] 4f1 5d 0 6s0
Therefore, it has only one unpaired electron. i.e., n = 1

Question. What is meant by ‘disproportionation’? Give an example of a disproportionation reaction in aqueous solution.
Answer.Disproportionation reaction involves the oxidation and reduction of the same substance. The examples of disproportionation reaction are :
(i) Aqueous NH3 when treated with Hg2Cl2 (solid)forms
mercury aminochloride disproportionatively.
Hg2Cl2 + 2NH3 → Hg + Hg(NH2)Cl + NH4Cl
(ii) 2Cu+ → Cu + Cu2+
Question. The magnetic moment of a transition metal ion is found to be 3.87 BM. How many number of unpaired electrons are present in it ?
Answer.
Magnetic moment, μeff = 3.87 BM corresponds to the number of unpaired electrons, n = 3 by applying the formula.
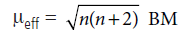
For n = 1, μ = 1.73 B.M, for n = 2, m = 2.83 BM
For n = 3, μ = 3.87 B.M and so on.
Question. How would you account for the following :
(i) Cr2+ is reducing in nature while with the same d-orbital configuration (d4), Mn3+ is an oxidising agent.
(ii) In a transition series of metals, the metal which exhibits the greatest number of oxidation states occurs in the middle of the series.
Answer.(i) E° values for the Cr3+/Cr2+ and Mn3+/Mn2+ couples are

These E° values indicate that Cr2+ is strongly reducing while Mn3+ is strongly oxidising agent.
(ii) Middle of the transition series contains greater number of unpaired electrons in (n –1)d and ns orbitals.
Question. Use the data to answer the following and also justify giving reasons :

(i) Which is a stronger reducing agent in aqueous medium, Cr2+ or Fe2+ and why?
(ii) Which is the most stable ion in +2 oxidation state and why?
Answer.
Cr2+ is a stronger reducing agent than Fe2+.
E°Cr3+/Cr2+ is negative (–0.41 V) whereas E°Fe3+/Fe2+ is positive (+ 0.77 V). Thus Cr2+ is easily oxidized to Cr3+ but Fe2+ cannot be easily oxidized to Fe3+. Hence, Cr2+ is stronger reducing agent than Fe2+.
(ii) More positive is the value of E°, reaction will be more feasible.
As E°Co3+/Co2+ is maximum, thus Co2+ ion is most stable.
Question. What is lanthanoid contraction and what is it due to? Write two consequences of lanthanoid contraction.
Answer.Lanthanoid contraction : The steady decrease in the atomic and ionic radii of lanthanoid elements with increase in atomic number is called lanthanoid contraction.
It is caused due to imperfect shielding of nuclear charge by 4f-electrons.
Consequences of lanthanoid contraction :
(i) The basic strength of oxides and hydroxides of lanthanoids decrease with increasing atomic number.
(ii) Atomic and ionic sizes of 4d transition series elements and 5d series elements are similar. e.g., atomic radii of zirconium(Zr) is same as that of hafnium (Hf).
Question. (i) Which metal in the first transition series
(3d-series) exhibits +1 oxidation state most frequently and why?
(ii) Which of following cations are coloured in aqueous solutions and why?
Sc3+, V3+, Ti4+, Mn2+
(At. Nos. Sc = 21, V = 23, Ti = 22, Mn = 25)
Answer.(i) Copper exhibits +1 oxidation state in its compounds.
Electronic configuration of Cu in the ground state is 3d10 4s1.
So, Cu can easily lose 4s1 electron to attain a stable 3d10 configuration. Thus, it shows +1 oxidation state.
(ii) Only those ions will be coloured which have partially filled d-orbitals facilitating d-d transition. Ions with d0 and d10 will be colourless.
From electronic configuration of the ions, V3+(3d2) and Mn2+(3d 5), are all coloured. Ti4+(3d 0) and Sc3+(3d0) are colourless.
Short Answer Type Questions (SA-II)
Question. How would you account for the following :
(i) The atomic radii of the metals of the third
(5d) series of transition elements are virtually the same as those of the corresponding members of the second (4d) series.
(ii) The E° value for the Mn3+/Mn2+ couple is much more positive than that for Fe3+/Fe2+ couple.
(iii) The highest oxidation state of a metal is exhibited in its oxide or fluoride.
Answer.(i) This is due to lanthanoid contraction.
(ii) Much larger third ionisation energy of Mn(where change is d5 to d4) is mainly responsible for this. This also explains that +3 state of Mn is of little importance.
From the relation, ΔG° = –nFE°
More positive is the value of E°, reaction will be feasible.

Hence, E°value for Mn3+/Mn2+ couple is much more positive than that for Fe3+/Fe2+.
(iii) Manganese can form pπ-dπ bond with oxygen by utilising 2p-orbital of oxygen and 3d-orbital of manganese due to which it can show highest oxidation state of +7. While with fluorine it cannot form such pπ – dπ bond thus, it can show a maximum oxidation state of +4.
Question. (i) Ce (IV) is a good analytical reagent. Why?
(ii) Account for the following : Copper(I)
compounds are white whereas copper(II)
compounds are coloured.
Answer.(i) The E° value for Ce4+/Ce3+ is 1.74 V which suggests that it can oxidise water. However the reaction rate is very slow and hence Ce (IV) is a good analytical reagent.
(ii) Cu(I) compounds have completely filled d-orbitals and there are no vacant d-orbitals for promotion of electrons whereas in Cu(II) compounds have one unpaired electron which is responsible for colour formation.
Question. Give reasons for the following :
(i) Mn3+ is a good oxidising agent.
(ii) EM2+/M values are not regular for first row transition metals (3d-series).
(iii) d-block elements exhibit more oxidation states than f-block elements.
Answer.(i) Mn2+ is more stable due to half filled d5 configuration and Mn3+ easily changes to Mn2+ hence, it is a good oxidising agent.
(ii) The E°M2+/M values are not regular which can be explained from the irregular variation of ionisation enthalpes
i.e., IE1 + IE2 and also the sublimation enthalpies which are relatively much less for manganese and vanadium.
(iii) All transition elements except the first and the last member in each series show a large number of variable oxidation states. This is because difference of energy in the
(n – 1)d and ns orbitals is very little.
Hence, electrons from both the energy levels can be used for bond formation.
Question. How do the oxides of transition elements in lower oxidation states differ from those in higher oxidation states and why?
Answer.In the lower oxidation state, the transition metal oxides are basic and they are acidic if the metal is in higher oxidation state. The oxides are amphoteric when the metal is in intermediate oxidation state.
For example,
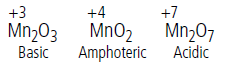
In case of lower oxide of a transition metal, the metal atom has a low oxidation state. This means some of the valence electrons of the metal atom are not involved in bonding,hence, these can be used for donation. Thus, these are act as bases.
Question. How would you account for the following :
(i) The oxidising power of oxoanions are in the order VO+2 < Cr2O72– < MnO4–
(ii) The third ionization enthalpy of manganese (Z = 25) is exceptionally high.
(iii) Cr2+ is a stronger reducing agent than Fe2+.
Answer.
(i) Change in Cr2O72– to Cr(III) is 3 and in MnO4– toMn (II) is 5.
Change in oxidation state is large and the stability of reduced products in V(III) < Cr(III) < Mn(II). This is why oxidising power of VO2+ < Cr2O72– < MnO4–.
(ii) Third ionization enthalpy of Mn is very high because the third electron has to be removed from the stable half-filled 3d-orbitals [Mn2+ (Z = 25) = 3d5].
(iii) Cr2+ is a stronger reducing agent than Fe2+. E°Cr3+/Cr2+ is negative (–0.41 V) whereas E°Fe3+/Fe2+ is positive (+ 0.77 V). Thus Cr2+ is easily oxidized to Cr3+ but Fe2+
cannot be easily oxidized to Fe3+. Hence, Cr2+ is stronger reducing agent than Fe2+.
Question. (i) Explain the cause of paramagnetism in lanthanoid ions.
(ii) Nb and Ta exhibit similar properties. Give reason.
(iii) Among the ionic species, Sc3+, Ce4+ and Eu2+,which one is a good oxidising agent.
Answer.(i) Due to presence of unpaired electrons in f-orbital lanthanoid ions are paramagnetic in nature.
(ii) Due to lanthanoid contraction, their sizes are same.
Hence, their properties are similar.
(iii) Ce4+. The stable oxidation state of lanthanoids is +3.Ce4+ tends to accept an electron to change to +3 state.
Hence, it acts as a good oxidising agent.
Question. (a) Assign reasons for the following :
(i) Cu(I) ion is not known to exist in aqueous solutions.
(ii) Transition metals are much harder than the alkali metals.
(b) Name the lanthanoids which show abnormally low value of third ionisation enthalpy.
Answer.(a) (i) In aqueous solutions, Cu+ undergoes disproportionation to form a more stable Cu2+ ion.
2Cu+(aq) → Cu2+(aq) + Cu(s)
Cu2+ in aqueous solutions is more stable than Cu+ ion because hydration enthalpy of Cu2+ is higher than that of Cu+. It compensates the second ionisation enthalpy of Cu involved in the formation of Cu2+ ions.
(ii) This is attributed to the involvement of greater number of electrons from (n –1)d in addition to the ns electrons in the interatomic metallic bonding.
(b) Third ionization enthalpy of lanthanoid is low if it leads to stable empty, half filled or completely filled configuration,as indicated by the abnormally low third ionization enthalpies of La, Gd, and Lu.
Question. Account for the following :
(i) The transition metals and their compounds act as good catalysts.
(ii) The lowest oxide of transition metal is basic, the highest is amphoteric/acidic.
(iii) The magnetic moment (B.M.) of Fe2+ ion is 24 .
Answer.(i) The transition metals and their compounds, are known for their catalytic activity. This activity is ascribed to their ability to adopt multiple oxidation states, ability to adsorb
the reactant(s) and ability to form complexes. Vanadium (V) oxide (in Contact Process), finely divided iron (in Haber’s Process), and nickel (in catalytic hydrogenation) are some of the examples.
(ii) Lowest oxidation compounds of transition metals are basic due to their ability to get oxidised to higher oxidation states.
Whereas, the higher oxidation state of metal and compounds gets reduced to lower ones and hence acts as acidic in nature. e.g., MnO is basic whereas Mn2O7 is acidic.
(iii) : Fe2+ ⇒ 3d 6, ∴ n = 4
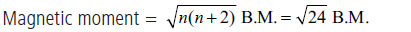
Question. Following are the transition metal ions of 3d series :
Ti4+, V2+, Mn3+, Cr3+
(Atomic numbers : Ti = 22, V = 23, Mn = 25, Cr = 24)
Answer the following :
(i) Which ion is most stable in aqueous solution and why?
(ii) Which ion is strong oxidising agent and why?
(iii) Which ion is colourless and why?
Answer.(i) Ti4+ has highest oxidation state among the given ions.
Ti4+ has stable inert gas configuration and hence, most stable in aqueous solution.
On the other hand, V2+, Mn3+, Cr3+ have unstable electronic configuration and hence, are less stable.
(ii) Due to presence of highest oxidation state of Ti, it acts as the strongest oxidising agent among the given ions.
(iii) Due to absence of unpaired electron in Ti4+, it is a colourless ion.
E.C. of Ti4+ : [Ar]3d 04s0
Question. Compare qualitatively the first and second ionisation potentials of copper and zinc. Explain the observation.
Answer.The electronic configuration of Zn and Cu are :
Zn: 1s2 2s2 2p6 3s2 3p6 3d10 4s2
Cu: 1s2 2s2 2p6 3s2 3p6 3d10 4s1
From the above configuration it is clear that first ionisation energy of Zn is greater than that of Cu (because of 4s2 and 4s1 configuration of Zn and Cu respectively). More energy is needed to remove an electron from 4s2 than from 4s1.
The second I.E. of Cu is higher than that of Zn because for Cu+ the configuration is 1s2 2s2 2p6 3s2 3p6 3d10 and for Zn+ the configuration is 1s2 2s2 2p6 3s2 3p6 3d10 4s1, it is easier to remove 4s1 electron of Zn+ than a 3d-electron from 3d10 (stable configuration).
Question. (a) Account the following :
(i) Transition metals form large number of complex compounds.
(ii) E°° value for the Mn3+/Mn2+ couple is highly positive (+1.57 V) as compared to Cr3+/Cr2+.
(iii) Which of following cations are coloured in aqueous solutions and why?
Sc3+, V3+, Ti4+, Mn2+
(At. Nos. Sc = 21, V = 23, Ti = 22, Mn = 25)
Answer.(a) (i) Transition metals form a large number of complex compounds due to following reasons :
– Comparatively smaller size of metal ions.
– High ionic charges.
– Availability of d-orbitals for bond formation.
(ii) Because Mn2+ is stable due to half filled configuration.
Thus Mn3+ has high tendency to form Mn2+ while Cr3+ is more stable than Cr2+.
(iii) Only those ions will be coloured which have partially filled d-orbitals facilitating d-d transition. Ions with d0 and d10 will be colourless.
From electronic configuration of the ions, V3+(3d2) and Mn2+(3d 5), are all coloured. Ti4+(3d 0) and Sc3+(3d 0) are colourless.
Question. (a) Explain the following :
The enthalpies of atomization of transition metals are quite high.
(b) Explain the following observations.
(i) With the same d-orbital configuration (d4), Cr2+ is a reducing agent while Mn3+ is an oxidising agent.
(ii) There is hardly any increase in atomic size with increasing atomic numbers in a series of transition metals.
Answer.(a) As transition metals have a large number of unpaired electrons in the d-orbitals of their atoms they have strong interatomic attraction or metallic bonds. Hence, they have high enthalpy of atomization.
(b) (i) E° values for the Cr3+/Cr2+ and Mn3+/Mn2+ couples are

These E° values indicate that Cr2+ is strongly reducing while Mn3+ is strongly oxidising agent.
(ii) As one proceeds along a transition series, the nuclear charge increases which tends to decrease the size but the addition of electrons in the d-subshell increases the screening effect which counterbalances the effect of increased nuclear charge. As a result, the atomic radii remain practically same after chromium.
Question. (i) Transition metals have very high melting and boiling points. Why?
(ii) In d-block element, ionic radii of ions of the same charge decreases progressively with increasing atomic number in a series. Why?
Answer.(i) The high melting and boiling points of transition metals are attributed to the involvement of greater number of electrons from (n – 1) d-orbital in addition to the ns electrons in the interatomic metallic bonding (d-d overlap).
(ii) As the atomic number increases the new electron enters the d-orbital and expected to increase in atomic size, but due to poor shielding effect of d-orbitals the electrostatic attraction between nucleus and outermost orbital increases and hence, the ionic radii decreases.
Question. (a)
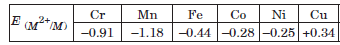
From the given data of E° values, answer the following questions :
(i) Why is E°(Cu2+/Cu) value exceptionally positive?
(ii) Why is E°(Mn2+/Mn) value highly negative as compared to other elements?
(b) Give reason and select one atom/ion which will exhibit asked property :
(i) Sc3+ or Cr3+ (exhibit diamagnetic behaviour)
(ii) Cr or Cu (high melting and boiling point)
Answer.(a) (i) Copper has high energy of atomisation and low energy of hydration.
(ii) Mn2+ ion has stable half-filled (3d5) electronic configuration.Its ionisation enthalpy value is lower in comparison to hydration enthalpy. Hence E °Mn2+/Mn is more negative.
(b) (i) Sc3+ has 3d 0 outer electronic configuration, therefore it is diamagnetic in nature whereas Cr3+ has 3d 3 outer electronic configuration. So, it is paramagnetic due to presence of unpaired electrons.
(ii) In a particular series, the metallic strength increases upto middle with increasing number of unpaired electrons,i.e., upto d5 configuration. After Cr, the number of unpaired
electrons goes on decreasing. Accordingly, the m.pt and b.pt. decrease after middle (Cr) because of increasing pairing of electrons.
Question. (i) Out of the ions Ag+, Co2+ and Ti4+ which will be coloured in aqueous solution?
(ii) If each one of the above ionic species is placed in a magnetic field, how will they respond and why?
Answer.
(i) The ionic species which possesses unpaired electron or electrons in (n – 1)d-subshell will show colour. Out of the ions Ag+(4d10), Co2+(3d7) and Ti4+(3d 0), Co2+ will be coloured as it contains three unpaired electrons, Ag+ andTi4+ will be colourless.
(ii) When placed in magnetic field, Co2+ will be attracted because it is paramagnetic due to unpaired electrons. Ag+ and Ti4+ ions will be repelled by the magnetic field as they are diamagnetic.
Long Answer Type Questions
Question. Compare the general characteristics of the first series of the transition metals with those of the second and third series metals in the respective vertical columns. Give special emphasis on the following points :
(i) electronic configuration
(ii) oxidation states
(iii) ionisation enthalpies and
(iv) atomic sizes.
Answer.(i) Electronic configuration : Electronic configuration of group 3 elements (Sc, Y, La) is [Noble gas]
(n – 1)d1 ns2.
Elements of group 6 (Cr, Mo and W) show exception in electronic configuration. For Cr and Mo [Noble gas] (n – 1)
d5 ns1 and for W it is [Noble gas] 4f 14 5d4 6s2.
Group 11 elements (Cu, Ag and Au) also show exceptional electronic configuration :
Cu : [Ar] 3d10 4s1, Ag : [Kr]4d10 5s1,
Au : [Xe]4f 14 5d 10 6s1.
Group – 10 (Ni, Pd and Pt) also show anomalous electronic configuration:
Ni : [Ar] 3d 8 4s2
Pd : [Kr] 4d10 5s0
Pt : [Xe] 4f 14 5d 9 6s1
(ii) Oxidation states : Elements within the same group show similar oxidation states. Highest number of oxidation states are shown by the elements lying in the middle of the transition series. Minimum oxidation states are shown by the elements lying near to left and far right side of the series.
Stability of higher oxidation states increases from first to third series.
(iii) Ionisation enthalpies : Ionisation enthalpies generally decrease down a group. This trend is followed from 3d to 4d-elements but the ionisation enthalpies either remain same or increase in going from 4d to 5d-series with the same group.This reverse trend is due to the poor shielding of the nuclear charge by the inner 4f-electrons. This increases the Zeff and in turn increases the ionisation enthalpy.
(iv) Atomic size : Due to poor shielding of nuclear charge by 4f-electrons, increase in Zeff decreases the size. So, the atomic size increase from 3d to 4d but decrease or remain almost the same from 4d to 5d.
Question. (a) Account for the following :
(i) Transition metals show variable oxidation states.
(ii) Zn, Cd and Hg are soft metals.
(b) Give reason :
Iron has higher enthalpy of atomization than that of copper.
(c) What are interstitial compounds? Write their properties.
Answer.
(a) (i) Transition elements can use their ns and (n – 1)d orbital electrons for bond formation therefore, they show variable oxidation states.
For example, Sc has ns2(n – 1) d1 electronic configuration.
It utilizes two electrons from its ns subshell then its oxidation state = +2. When it utilizes both the electrons then its oxidation state = +3.
(ii) In Zn, Cd and Hg, all the electrons in d-subshell are paired. Hence, the metallic bonds are weak. That is why they are soft metals with low melting and boiling points.
(b) Greater the number of unpaired electrons, stronger is the metallic bond and therefore, higher is the enthalpy of atomisation. Since, iron has greater number of unpaired
electrons than copper hence has higher enthalpy of atomisation.
(c) When small atoms of non metals like H, C, B, N etc can occupy vacant interstitial spaces in condition metals it give size to interstitial compound like hydrides, carbides. Few properties are as follow
(i) They have high melting points then pure metals.
(ii) They are conductive.
(iii) They are chemically inert.
Question. (a) The elements of 3d transition series are given as Sc Ti V Cr Mn Fe Co Ni Cu Zn
Answer the following :
(i) Write the element which shows maximum number of oxidation states. Given reason.
(ii) Which element has the highest melting point?
(iii) Which element shows only +3 oxidation state?
(iv) Which element is a strong oxidising agent in +3 oxidation state and why?
(v) Why Mn2O3 is basic whereas Mn2O7 is acidic?
Answer.(i) Mn shows maximum no. of oxidation states from +2 to +7 because Mn has maximum number of unpaired electrons in 3d sub-shell.
(ii) Cr has maximum melting point, because it has 6 unpaired electrons in the valence shell, hence it has strong interatomic interaction.
(iii) Sc shows only +3 oxidation state because after losing 3 electrons, it has noble gas electronic configuration.
(iv) Mn is strong oxidising agent in +3 oxidation state because change of Mn3+ to Mn2+ give stable half filled (d 5) electronic configuration,E°(Mn3+/Mn2+) = 1.5 V.
(v) Basic nature of oxides decreases and acidic nature increases with increase in oxidation state of the metal.
Oxidation state of Mn in Mn2O3 is +3 while in Mn2O7 is +7.
Question. Give reasons for the following :
(i) Silver bromide is used in photography.
(ii) Most transition metal compounds are coloured.
(iii) Zinc and not copper is used for the recovery of metallic silver from complex [Ag(CN)2]–. Explain.
(iv) The colour of mercurous chloride, Hg2Cl2,changes from white to black when treated with ammonia.
(v) The species [CuCl4]2– exists while [CuI4]2– does not.
Answer.
(i) Silver bromide is used in photography because of its sensitivity to sunlight. In light, AgBr reduces to metallic silver.
(ii) The colour of transition metal compound is due to the presence of incompletely filled d-orbitals in transition metal ions/atoms, because of this d-d transition can occur in them. The colour is due to d-d transition for which the energy is absorbed from visible region. The visible colour of a compound is the complementary colour of the absorbed light.
(iii) Zinc is a cheaper and stronger reducing agent as compared to copper.
(iv) Mercurous chloride (white) changes to black on treatment with ammonia because of the formation of finely divided mercury (grey).
(v) Cu2+ is reduced to Cu+ by I– and thus CuI2 gets converted to Cu2I2. This change cannot be brought about by Cl–.
Question. (a) Give reason :
(i) Sc (21) is a transition element but Ca (20) is not.
(ii) The Fe2+ is much more easily oxidised to Fe3+ than Mn2+ to Mn3+.
(b) How would you account for the following :
(i) Metal-metal bonding is more extensive in the 4d and 5d series of transition elements than the 3d series.
(ii) Mn (III) undergoes disproportionation reaction easily.
(iii) Co (II) is easily oxidised in the presence of strong ligands.
Answer.(a) (i) Sc(21) is a transition element but Ca(20) is not because of incompletely filled 3d orbitals.
(ii) Electronic configuration of Mn2+ is 3d 5 which is half filled and hence stable. Therefore, third ionization enthalpy is very high, i.e., 3rd electron cannot be lost easily. In case of Fe2+, electronic configuration is 3d 6. Hence, it can lose one electron easily to give the stable configuration 3d 5.
(b) (i) The metals of 4d and 5d-series have more frequent metal bonding in their compounds than the 3d-metals because 4d and 5d-orbitals are more exposed in space than the 3d-orbitals. So the valence electrons are less tightly held and form metal-metal bonding more frequently.
(ii) Mn3+ is less stable and changes to Mn2+ which is more stable due to half-filled d-orbital configuration. That is why, Mn3+ undergoes disproportionation reaction.
(iii) The tendency to form complexes is high for Co(III) as compared to Co(II). Co2+ ions are very stable and are difficult to oxidise. Co3+ ions are less stable and are reduced by water.
In contrast many Co(II) complexes are readily oxidised to Co(III) complexes and Co(III) complexes are very stable, e.g.,

This happens because the crystal field stabilisation energy of Co(III) with a d 6(t26g) configuration is higher than for Co(II) with a d7 (t 6 2g eg1) arrangement.
